 
Research and clinical efforts for other conditions. Personalized medicine in orphan diseases, that could ultimately aid in similar Genetic profiles, narcolepsy may provide a nice example for developing Given the different clinical, biological and Further well-designedĬontrolled trials are required to determine if they could really impact on the Possible after disease onset could theoretically slow down or stop theĭestruction of ORX neurons in some selected patients. In addition, immune-based therapies administered as early as NT1 opened new therapeutic options oriented towards ORX-based therapies,Įspecially nonpeptide ORX receptor agonists that are currently underĭevelopment. Sodium oxybate, usedįor decades in adult patients with narcolepsy, was recently shown to beĮffective and safe in childhood narcolepsy. Soon a new therapeutic option to treat EDS in NT1 and NT2. Phenylalanine derivative with dopaminergic and noradrenergic activity will be Recently approved to treat patients with NT1 and NT2. Pitolisant, a selective histamine H3 receptor inverse agonist, has been Narcolepsy is an active area for drugĭevelopment, and new wake-promoting agents have been developed over the past With psychostimulants, as modafinil/armodafinil, methylphenidate, orĪmphetamines as a third-line therapy. 
The main and most disabling symptom, EDS, is managed 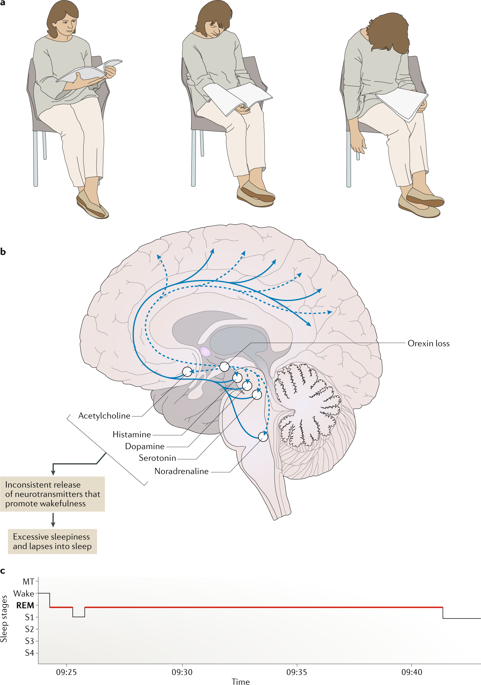
Our understanding of the neurobiological basis of NT1, management remains Sleep, but without cataplexy and loss of ORX neurons. Little is known about NT2 etiology and prevalence, sharing with NT1Įxcessive daytime sleepiness (EDS) and dysregulation of rapid eye movement (REM) Irreversible loss of hypocretin/orexin (ORX) neurons, by a probable autoimmune Emerging treatments are under development newly developed wakefulness-promoting agents act via different mechanisms of action, whereas other forms of therapy are focused in the underlying hypocretin deficiency that characterizes narcolepsy type 1.Įxcessive daytime sleepiness Hypersomnolence Narcolepsy Stimulant Wakefulness-promoting agent.Narcolepsy type 1 (NT1) is a chronic orphan disorder, caused by the selective and Additionally to the wakefulness-promoting agents and stimulants classically used, other pharmacologic options have been recently approved for the treatment of EDS in Europe (pitolisant) and the US (solriamfetol). Ongoing research of new molecules is based on several mechanisms of action (histamine antagonists/inverse agonists, GABA receptor modulators), and potential future strategies involve immunologic treatment and hypocretin-based therapies. Further studies involving the paediatric population are warranted to have solid evidence in the management of children with narcolepsy. Sodium oxybate was recently approved for EDS management in paediatric patients from the age of seven. More recently, pitolisant (H3 receptor inverse agonist) was approved by the European Medicines Agency, and solriamfetol (dopamine and norepinephrine reuptake inhibitor) by the Food and Drug Administration, for the treatment of EDS in adult narcolepsy patients. Other options with a stimulant effect, such as methylphenidate and amphetamines are considered if the former do not control the symptoms. An overview of novel therapies and potential future options are covered as well.įirst line treatments for EDS in narcolepsy patients include modafinil/armodafinil and sodium oxybate. The aim of this review is to discuss and summarize the main therapeutic strategies for the management of excessive daytime sleepiness (EDS) in patients with narcolepsy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
